Recommended composition of influenza virus vaccines for use in the 2020 - 2021 northern hemisphere influenza season 28 February 2020. The vaccine is marketed as Fluad in the US and was first available in the 20162017 flu season.
 Fluzone High Dose Influenza Vaccine Sanofi Flu
Fluzone High Dose Influenza Vaccine Sanofi Flu
Are quadrivalent flu vaccines safe.
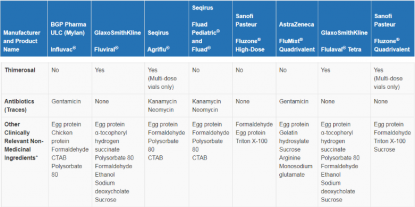
Quadrivalent flu vaccine ingredients. In 20202021 this is one known as the Phuket strain. For active immunization for the prevention of influenza disease caused by influenza. For active immunization for the prevention of influenza disease caused by influenza virus subtypes A and type B viruses contained in the vaccine.
2 DOSAGE AND ADMINISTRATION For intramuscular injection only. For individuals 6 months through 17 years of age. Most of the time it takes a few weeks for Flublok Quadrivalent influenza virus vaccine recombinant to work.
The ingredient called adjuvant MF59 is a natural compound that can be made from vegetable oils or more commonly shark liver oil. People who get influenza may have a fever chills cough runny nose sore throat headache muscle aches extreme weakness and fatigue. It is the first adjuvanted seasonal flu vaccine marketed in the United States.
Fluarix Quadrivalent is prepared from influenza viruses propagated in embryonated chicken eggs. FLUAD QUADRIVALENT is an active immunization of persons 65 years of age and older against influenza disease caused by seasonal influenza virus subtypes A and types B. You should receive a flu vaccine every year.
It is recommended that quadrivalent vaccines for use in the 2020 - 2021 northern hemisphere influenza season contain the following. AGuangdong-MaonanSWL15362019 CNIC-1909 H1N1 AHong Kong26712019 IVR-208 H3N2 BPhuket30732013 B Yamagata lineage and BWashington022019 B Victoria lineage. Fluarix PF Quadrivalent 2020-2021 is used to prevent infection caused by influenza virus.
Your immunity will gradually decrease over the 12 months after you receive the Fluzone Quadrivalent 2020-2021. The vaccine is redeveloped each year to contain specific strains of inactivated killed flu virus that are recommended by public health officials for that year. Fluarix Quadrivalent Influenza Vaccine for intramuscular injection is a sterile colorless and slightly opalescent suspension.
FLUAD QUADRIVALENT is an inactivated influenza vaccine indicated for active immunization against influenza disease caused by influenza virus subtypes A and types B contained in the vaccine. What is influenza. Most flu vaccine in the United States is now quadrivalent.
21 Dosage and Schedule The dose and schedule for FLUARIX QUADRIVALENT are presented in Table 1. Children receiving this vaccine may need a booster shot one month after receiving the first vaccine. Fluzone High-Dose Quadrivalent is standardized according to United States Public Health Service requirements and is formulated to contain HA of each of the following four influenza strains recommended for the 2020-2021 influenza season.
This new vaccine contains an extra ingredient called an adjuvant that promotes a stronger immune response to the flu vaccine. For those who do get sick after getting a flu shot vaccination may reduce. Product Information Package Insert - FluLaval Quadrivalent.
For active immunization for the prevention of disease caused by influenza A subtype viruses and type B viruses contained in the vaccine. The safety of Quadrivalent Influenza Vaccine split virion inactivated was assessed in six clinical trials in which 3040 adults from 18 to 60 years of age 1392 elderly over 60 years of age and 429 children from 9 to 17 years of age received one dose of Quadrivalent Influenza Vaccine split virion inactivated and 884 children from 3 to 8. A person can also get a quadrivalent or four-component vaccine that protects against an additional influenza B virus.
FLUARIX QUADRIVALENT is approved for use in persons aged 6 months and older. It must be given before you are exposed to the flu in order to work. Influenza is an acute respiratory illness that is caused by a virus.
Quadrivalent Influenza Vaccine split virion inactivated suspension for injection in pre-filled syringe - Patient Information Leaflet PIL by Sanofi Pasteur. Of the 194 to 198 million doses of flu vaccine projected to be available for the 2020-2021 season manufacturers estimate that 99 of doses 195 million will be quadrivalent flu vaccine. The flu vaccine is about 40 to 60 percent effective when the virus type in the vaccine aligns with the circulating virus.
The Fluzone Quadrivalent 2020-2021 is usually given in October or November. By influenza A subtype viruses and type B viruses contained in the vaccine see Description 11. The vaccine contains the MF59C1 adjuvant which is an oil-in-water emulsion of squalene oil.
This medicine is a vaccine made with proteins that cannot cause the flu. Each of the influenza viruses is produced and purified separately. This medicine is not a cure for the flu.
Cari Blog Ini
Arsip Blog
- Februari 2021 (18)
- Desember 2019 (3)
- November 2019 (5)
- Oktober 2019 (6)
- Agustus 2019 (10)
- Juli 2019 (30)
- Juni 2019 (176)
- Mei 2019 (25)
- Oktober 2018 (43)
- September 2018 (30)
- Agustus 2018 (58)
- Juli 2018 (67)
- Juni 2018 (238)
- Mei 2018 (41)
- Maret 2018 (50)
- Februari 2018 (107)
- Januari 2018 (70)
- Oktober 2017 (57)
- Agustus 2017 (78)
- Mei 2017 (39)
- April 2017 (28)
- Februari 2017 (13)
- Januari 2017 (134)
- Desember 2016 (297)
- November 2016 (1003)
- Oktober 2016 (360)
Label
- 1000an
- 1080p
- 12th
- 16x9
- 1920x1080
- 1969
- 2010
- 2013
- 2015
- 2016
- 2017
- 2018
- 2019
- 2020
- 2021
- 2077
- 23rd
- 2560x1440
- 30th
- 350v2
- 3840x2160
- 40th
- 44th
- 4kwalpapers
- 60x60
- 60x80
- 7zip
- about
- abstract
- accent
- accord
- accounting
- acoustic
- across
- acrylic
- activity
- actress
- adam
- adding
- addition
- address
- adenovirus
- adhesive
- adidas
- adjustments
- adults
- aesthetic
- africa
- african
- afrocentric
- after
- against
- agency
- agent
- agreement
- aids
- airplane
- airpod
- album
- alchemy
- alcohol
- algebra
- aliens
- allergy
- alphabet
- aluminum
- amazing
- amazon
- ambe
- america
- american
- amica
- ampela
- anaglypta
- anak
- anatomy
- android
- aneka
- animal
- animals
- animated
- anime
- ankle
- anne
- anniversary
- another
- answering
- answers
- anti
- aplia
- apple
- application
- aquarium
- architect
- arctic
- area
- army
- arrow
- artists
- artmax
- asam
- asem
- asin
- asinan
- asleep
- asli
- assemble
- assertive
- atletico
- atom
- attorney
- attractive
- audi
- aurora
- australia
- australian
- authentic
- auto
- autumn
- avengers
- aviators
- away
- awesome
- ayam
- baba
- bababooey
- babies
- baby
- back
- background
- backgrounds
- backsplash
- backwards
- backyard
- badge
- bagiak
- bahan
- bahasa
- bailey
- bajongko
- bakpao
- bakso
- balado
- balaji
- ball
- balls
- banana
- band
- bandeng
- bands
- bandung
- banff
- bangka
- barbie
- barcelona
- bargain
- barn
- barrel
- basic
- basil
- basket
- bass
- bassett
- bassinet
- bath
- bathroom
- batik
- batman
- battle
- bawang
- bday
- beach
- bead
- bean
- beans
- bear
- beard
- beatitudes
- beatrice
- beautiful
- beauty
- bebek
- become
- bedroom
- beef
- beginner
- beginners
- beginning
- bein
- bekal
- beku
- believes
- bening
- bento
- beras
- berbahan
- berk
- best
- betawi
- better
- bhagwan
- bible
- bicicletas
- bicycle
- bieber
- bihun
- biji
- bike
- bikes
- bikin
- bill
- binod
- birch
- bird
- birds
- birthday
- biskuit
- black
- blend
- blender
- blessings
- blood
- bloopers
- blossom
- blossoms
- blown
- blue
- bluetooth
- blum
- blush
- boat
- bobber
- bobby
- boho
- bollywood
- bolu
- book
- bookrunner
- books
- boost
- booster
- border
- borders
- borealis
- boston
- botanical
- bots
- bottle
- boxing
- boyfriend
- boys
- brain
- brand
- bras
- brazilian
- breaking
- breathable
- breed
- breeding
- brew
- brick
- bridal
- bride
- bridge
- bright
- bronco
- brown
- brownies
- bruins
- brushed
- bryant
- buah
- buat
- buddha
- budget
- buff
- building
- bulan
- bulb
- bumbu
- bunny
- burger
- burnout
- business
- butter
- butterfly
- button
- cabe
- cabin
- cable
- cactus
- cakalang
- cake
- cakes
- cakwe
- calculator
- calendar
- calendrier
- california
- call
- called
- calligraphy
- camaro
- camera
- camo
- campur
- canada
- canai
- cancer
- candy
- cane
- canvas
- capitan
- caps
- captain
- card
- cards
- care
- caribbean
- cars
- cartoon
- case
- cashman
- cast
- castle
- catch
- cbr1000rr
- cell
- cells
- celtic
- cemilan
- central
- cepat
- ceramic
- certificates
- cetaphil
- chair
- chakras
- chalk
- challenging
- chanel
- change
- chaplet
- chapter
- characteristics
- charger
- charlotte
- chart
- cheap
- cheat
- cheater
- cheats
- check
- cheese
- chegg
- chek
- chemicals
- cherry
- chess
- chicken
- chickenpox
- chihuahua
- chikungunya
- child
- childhood
- childrens
- china
- chinese
- chip
- chips
- chivas
- chocolate
- christian
- christmas
- chroma
- chromebook
- cilok
- cincane
- circle
- cite
- citizens
- city
- civic
- claire
- class
- classical
- classy
- clean
- clear
- client
- climate
- clinic
- clip
- clipart
- clips
- clock
- close
- cloth
- clothes
- cloud
- cloudy
- club
- clube
- cobain
- coco
- code
- coffee
- coklat
- cola
- cold
- collage
- college
- cologne
- color
- colorado
- colorful
- coloring
- colour
- colourful
- column
- combiner
- comic
- comics
- commandments
- commentators
- commercial
- common
- community
- compact
- companies
- company
- compensation
- completed
- comprehension
- computer
- computers
- concealment
- cone
- confections
- connect
- consolidation
- consonants
- construction
- contain
- control
- controlled
- controller
- convert
- cook
- cookies
- cool
- cooler
- coop
- copper
- copyright
- core
- corgi
- cornavirus
- coronavirus
- corps
- correct
- corvette
- cosmos
- cosplay
- cost
- costs
- costume
- couch
- cough
- council
- country
- couple
- couples
- coupon
- coupons
- court
- cover
- coverage
- covering
- covid
- crayons
- crazy
- cream
- creamy
- create
- creative
- creator
- creature
- crepes
- crest
- cricket
- cricut
- crispy
- critical
- crochet
- crossing
- crouser
- cryptocurrency
- crystal
- cuka
- cumbia
- cumi
- cura
- curry
- cursive
- custody
- custom
- customer
- cute
- cyberpunk
- cyclones
- cynthia
- dadar
- daging
- dallas
- damask
- dame
- dance
- dancing
- dank
- dara
- dari
- dark
- dasar
- date
- daughters
- dazed
- deadpool
- deals
- dean
- deaths
- debm
- debt
- decals
- decimal
- decimals
- deck
- decks
- declaration
- decor
- decorating
- decoration
- decorations
- decorative
- deed
- definition
- definitions
- degree
- deku
- delivery
- dello
- demon
- dental
- department
- deportivo
- deposit
- descendants
- desert
- design
- designer
- designers
- designs
- desk
- desktop
- dessert
- develop
- devi
- device
- devices
- dewasa
- diana
- diapers
- diego
- diet
- difference
- different
- digital
- dijual
- dimensions
- dimsum
- dining
- diphtheria
- disaster
- discord
- discount
- disney
- distemper
- distress
- distressed
- dividing
- division
- doctor
- doctors
- does
- doggo
- dogs
- dollars
- dolphin
- dont
- dots
- double
- doubling
- download
- downloadable
- downloads
- dragon
- dratini
- draw
- drawing
- dress
- dresses
- drip
- driver
- driving
- drumm
- drunk
- dslr
- ducks
- dulhan
- dumpling
- dumpster
- dunkin
- durga
- durian
- duty
- earbuds
- earlier
- earrings
- east
- easy
- economics
- edition
- eevee
- effect
- effective
- effects
- elastique
- election
- electronic
- elegant
- elementary
- elements
- elephant
- elizabeth
- ellie
- elvis
- emerald
- emoji
- emojis
- empek
- employment
- empty
- emulator
- enak
- enchanted
- enforcement
- engagement
- english
- enterprise
- entries
- episode
- escape
- espresso
- essential
- estrada
- eternatus
- ethernet
- etsy
- europe
- evaluation
- events
- everglades
- evoque
- example
- excel
- excellence
- exclusives
- exercise
- exercises
- exit
- expansion
- extension
- face
- facetime
- factory
- facts
- fairy
- faith
- fake
- fall
- Family
- Family Tree Chart Maker Online Free
- famous
- farm
- farmers
- farmhouse
- farrow
- fast
- faux
- feed
- feline
- felv
- female
- fence
- fern
- ferrell
- field
- fifty
- figures
- file
- files
- fillet
- film
- filter
- final
- financial
- financially
- find
- finding
- finger
- fingers
- fire
- first
- fish
- fitness
- fixtures
- flag
- flamengo
- flamingo
- fletcher
- flights
- flooring
- floors
- floral
- florida
- floss
- flower
- flowers
- flue
- fluminense
- fluvirin
- fogging
- fondant
- food
- foot
- footage
- football
- ford
- forest
- fork
- form
- format
- forms
- fortnite
- foto
- four
- fraction
- fractions
- framed
- frames
- francisco
- free
- freeze
- frequency
- fresh
- friday
- fridge
- friend
- friends
- from
- front
- frosting
- fruit
- fudgy
- full
- fund
- funny
- fuyunghai
- gaara
- gabus
- galaxy
- gallery
- gallo
- galvanized
- gambar
- game
- games
- gaming
- ganesh
- garage
- garden
- gardens
- gates
- gazzetta
- gear
- generator
- geometric
- george
- geprek
- gets
- ghibli
- ghoul
- gifs
- gift
- gifts
- gigabyte
- giovanni
- giraffe
- girl
- girlfriend
- girls
- girly
- glass
- glasses
- glitter
- glossy
- gochujang
- godog
- gogh
- gold
- golf
- good
- goods
- gopro
- gore
- goreng
- gori
- gotcha
- government
- gown
- gowns
- goyard
- grade
- graders
- gradient
- graffiti
- graphic
- graphite
- grasscloth
- grassland
- gratis
- grau
- gray
- great
- green
- grey
- grill
- groundhog
- gt500
- gudeg
- guitar
- gulab
- gulai
- gulung
- gurih
- hack
- hair
- halen
- half
- halloween
- hamil
- handayani
- handle
- handlebars
- hands
- handwriting
- hang
- hanging
- hangings
- hanuman
- happy
- hari
- harley
- harry
- hashtags
- hatching
- have
- headboard
- headphones
- health
- healthy
- hear
- heart
- hebrew
- heels
- hello
- hepatitis
- hero
- heroes
- heroine
- heron
- hexagon
- hgtv
- hidden
- hide
- high
- highest
- highland
- highlights
- hindi
- hippo
- History
- hobby
- hocks
- hogwarts
- hold
- holder
- hollywood
- holt
- home
- homeowners
- homework
- honda
- hondata
- honeycomb
- hoodie
- horse
- horses
- hotel
- hour
- howard
- hubble
- huge
- human
- humana
- hunter
- husband
- hydrate
- icon
- ideas
- igtv
- ikan
- ikonik
- image
- images
- imajinasi
- improve
- incapto
- inch
- increase
- india
- indian
- indonesia
- indoor
- industrial
- infinity
- influenza
- information
- ingkung
- ingles
- ingredients
- initiative
- injury
- inkay
- insect
- inserts
- inspiration
- inspirational
- install
- instant
- insurance
- integrity
- intercept
- interior
- international
- into
- investor
- invitation
- invitations
- iphone
- ireland
- irish
- iron
- islamic
- ivory
- iwatch
- jacks
- jadul
- jagger
- jagung
- jalangkote
- james
- jangan
- japanese
- jeans
- jeep
- jellycat
- jengkol
- jepang
- jesus
- jogja
- joined
- jordan
- josephine
- jualan
- jungkook
- jungle
- justice
- justin
- kacang
- kakap
- kakkar
- kale
- kambing
- kamboja
- kamir
- kampung
- kangaskhan
- karage
- kare
- kastengel
- katsu
- kawaii
- kebuli
- keju
- kekinian
- kelapa
- kemangi
- kembang
- kendrick
- kentang
- kepala
- kerang
- keren
- kering
- kerupuk
- ketan
- keto
- keurig
- keyboard
- khali
- khas
- kids
- killed
- kindergarten
- king
- kingdom
- kitchen
- kitten
- kittens
- kitty
- klepon
- knitting
- knockoff
- kobe
- konidela
- kopi
- krishna
- krispi
- kroket
- kuah
- kucing
- kudus
- kukus
- kuliner
- kulit
- kuning
- kupang
- kurt
- label
- labu
- ladies
- lakes
- lakh
- lakshmi
- lamar
- lamp
- landscape
- landscapes
- landscaping
- lanka
- lapis
- laptop
- large
- late
- latest
- laughing
- laws
- laxmi
- layer
- leader
- leaderboards
- leaf
- league
- lease
- leaves
- lebron
- leches
- legendary
- legs
- lembut
- lenggang
- lengkuas
- lepto
- less
- letter
- letters
- leukemia
- level
- levels
- liability
- liberty
- license
- lidah
- life
- lifestyle
- light
- lighting
- lightning
- lights
- lilly
- lily
- linen
- lines
- link
- lion
- list
- listen
- little
- live
- liver
- liverpool
- living
- liwet
- lobby
- location
- lodge
- login
- logo
- long
- longhorn
- look
- looking
- lookup
- looney
- loops
- lord
- love
- lovers
- lowercase
- lsvt
- luciferase
- luckin
- luigia
- lumer
- luxury
- lyrics
- machine
- mackinac
- madeline
- madrid
- madura
- magnetic
- magnolia
- mahalakshmi
- maiden
- maintenance
- maizena
- makan
- makanan
- makaroni
- make
- maker
- makeup
- malayalam
- malaysia
- male
- mamoswine
- managerial
- mandala
- mandhi
- manger
- mangga
- mango
- mangut
- manhattan
- manis
- manual
- manufactured
- many
- marble
- march
- marilyn
- marine
- market
- marketing
- married
- marseille
- martabak
- marvel
- masak
- masakan
- mashed
- mask
- master
- mata
- matah
- match
- math
- maths
- mati
- matted
- maxi
- mayonaise
- mcgregor
- mclaren
- mcqueen
- mcv4
- meaning
- measles
- medallion
- medan
- medical
- medicare
- meditation
- mega
- mehandi
- mekar
- membership
- membuat
- meme
- memes
- memorize
- mendoan
- meningitis
- meningococcal
- mens
- mentega
- menu
- mercedes
- merck
- mercon
- mercy
- meriah
- merit
- merry
- meta
- metal
- metallic
- michaels
- microsoft
- middle
- mille
- minecraft
- mini
- miniature
- minimalist
- minion
- minions
- minuman
- mistake
- mixed
- mobile
- modeling
- modern
- moderna
- moles
- monalisa
- monday
- money
- monica
- monkey
- mono
- monochrome
- monster
- montana
- monthly
- moon
- more
- morning
- morty
- mosaic
- moss
- most
- mother
- motherboard
- motion
- motocross
- motor
- motorcycles
- mountain
- mousse
- move
- moveset
- movie
- moving
- mpasi
- much
- mudah
- mudcloth
- mugs
- mujair
- mulch
- multi
- multiple
- multiplication
- mundo
- mung
- murah
- mural
- murals
- murder
- Music
- Musical
- mustang
- mutant
- Mystery
- nachrichten
- name
- names
- nangka
- nanoparticles
- narrow
- nasi
- nastar
- national
- nativity
- natural
- nature
- navratri
- navy
- near
- neck
- need
- negotiate
- neha
- neon
- nest
- nests
- netflix
- neutral
- newborn
- News
- newsprint
- nice
- nigeria
- night
- nike
- ninja
- nivea
- note
- number
- numbers
- numer
- nursery
- oatmeal
- ocean
- octopus
- odading
- office
- officiant
- often
- ohio
- olahan
- older
- olds
- online
- Online Gantt Chart Maker Free
- onyx
- open
- opener
- operations
- opiniones
- opor
- order
- ordering
- oregon
- oreo
- Organizational Chart Maker Free Online
- original
- ornaments
- oseng
- otter
- outbreak
- outdoor
- oven
- over
- oversized
- oxford
- padang
- pages
- pagina
- pahlawan
- paid
- pain
- paint
- painting
- paintings
- paints
- palace
- palembang
- palette
- palm
- pancake
- panchmukhi
- panda
- pandausb
- panel
- panggang
- panoramic
- panther
- pantone
- paper
- papillon
- paprika
- para
- paragon
- paralysis
- paras
- pari
- paris
- parivar
- party
- paso
- pass
- password
- pastel
- pathophysiology
- patin
- patrol
- patron
- patterns
- pattinson
- paving
- payoff
- pcv7
- pears
- peda
- pediatrician
- peel
- pekalongan
- pempek
- pencil
- penkek
- pension
- pentel
- pentol
- peony
- people
- pepaya
- pepes
- percent
- percents
- perdendo
- perfume
- periodic
- perk
- permanent
- person
- personalized
- pertussis
- pesmol
- petis
- pexels
- peyek
- pfizer
- phases
- philippines
- philosophy
- phone
- phones
- phonics
- phool
- photo
- photography
- photos
- photoshoot
- photoshop
- phrase
- piche
- picher
- pics
- picture
- pictures
- piece
- pier
- pikachu
- pill
- pindang
- pine
- pineapple
- pink
- pipe
- pirate
- pirex
- pisang
- piscok
- pitcher
- pittsburgh
- place
- plan
- planner
- plans
- plant
- plasma
- play
- playboy
- players
- playr
- playroom
- playstation
- plugs
- plum
- plus
- pods
- pokedex
- pokemon
- police
- policy
- polio
- politik
- polka
- polo
- pool
- popular
- porsche
- portable
- portrait
- positive
- poster
- posters
- potato
- pottery
- pour
- powerstroke
- practice
- practive
- praktis
- pray
- premier
- preschool
- preschoolers
- prevent
- preventive
- prevnar
- price
- prices
- prima
- primer
- princess
- printable
- printables
- printed
- printer
- prints
- problem
- problems
- proctored
- professional
- profile
- program
- progressive
- project
- promote
- prompts
- proofreading
- propane
- property
- pros
- psychologist
- puding
- pulitzer
- pulls
- pulsar
- puma
- pump
- pumpkins
- pumps
- puppies
- puppy
- purple
- putih
- puyuh
- quadrille
- quadrivalent
- quality
- queen
- quest
- questions
- queue
- quilted
- quote
- quotes
- rabies
- racing
- radha
- radio
- raid
- raiders
- rainbow
- rainforest
- raise
- raised
- rakhi
- rallye
- randomizer
- range
- ranger
- rarest
- ratios
- rayner
- reactions
- reading
- real
- rebel
- reception
- recife
- recipe
- reconciliation
- reconstruction
- records
- recover
- rectangle
- redskins
- regal
- regina
- register
- registry
- regrouping
- regulations
- rehabilitation
- relay
- release
- remainder
- remote
- removable
- remove
- rendang
- rendering
- renesans
- rent
- rental
- renyah
- replace
- replacing
- repsol
- resep
- reset
- resized
- resmi
- resolution
- resources
- response
- retro
- returns
- reusable
- review
- reviews
- richard
- rick
- ring
- rings
- ripe
- rising
- rivera
- road
- roadrunner
- roasters
- robert
- roblox
- rock
- rogue
- rolade
- roleplaying
- roll
- rollout
- romans
- ronaldo
- roof
- room
- rose
- roses
- roti
- round
- rouser
- rover
- royal
- royalty
- rugby
- rujak
- rule
- rules
- rumahan
- rust
- rustic
- sabbath
- safavieh
- safety
- saggy
- sagon
- sahur
- said
- saints
- sajiku
- sakura
- salad
- salak
- salary
- sale
- sales
- salk
- salle
- salvation
- sambal
- sample
- sams
- samsung
- sand
- sandler
- sanitizer
- santa
- santan
- santorini
- saori
- sapi
- saraswati
- sarden
- sate
- saudara
- saus
- saying
- sayur
- scar
- scene
- scenery
- scenes
- schedule
- scheduler
- schnauzer
- science
- scientific
- sconce
- score
- screen
- screensaver
- screensavers
- script
- scripture
- sculpture
- scutes
- seahawks
- search
- seascape
- season
- Seating Chart Maker Free Online
- seats
- seattle
- seblak
- sebulan
- second
- section
- sederhana
- sehat
- selai
- selected
- self
- sell
- sells
- semarang
- semprit
- semur
- sentences
- sequential
- serabi
- serani
- series
- serundeng
- servers
- service
- setan
- sets
- setting
- shabbat
- shadow
- shallots
- shapes
- shark
- sheet
- shelby
- shelf
- shibori
- shield
- shingles
- shingrix
- ship
- shipping
- shirt
- shirts
- shiva
- shoes
- shopping
- shore
- shot
- shots
- shower
- shree
- shri
- siang
- side
- sight
- sign
- signature
- sikkim
- silky
- silver
- simple
- simpson
- sing
- singer
- singkong
- sinker
- siomay
- sister
- sites
- sixth
- size
- sizer
- skills
- skin
- skinny
- skull
- skyline
- slash
- slayer
- sleep
- slip
- slow
- small
- smart
- smartphone
- smile
- smoke
- smoker
- smoothies
- snapchat
- sneaker
- snoopy
- snow
- snowboard
- snowflake
- snowman
- sobek
- sobro
- soccer
- social
- software
- sokaraja
- solo
- someone
- song
- songs
- sonntag
- sony
- sosis
- soto
- souffle
- sound
- source
- south
- southern
- space
- spanish
- speakers
- spell
- spelling
- spencer
- spesial
- spinach
- spirit
- splash
- split
- spongebob
- spoon
- Sport
- sportback
- sporting
- sports
- spreadsheet
- spring
- sprout
- srikaya
- stadium
- stain
- stains
- stand
- standing
- star
- starbucks
- starfleet
- stargazer
- starry
- starters
- state
- static
- statue
- status
- steak
- steakhouse
- steamboat
- steelers
- stick
- stickable
- sticker
- stickers
- stilettos
- still
- stingray
- stock
- stockimagebank
- stone
- storage
- store
- storm
- story
- straberries
- stratton
- stream
- streaming
- stretch
- string
- strings
- student
- students
- studies
- studio
- stuffed
- style
- styles
- stylish
- sublimation
- subscription
- subtraction
- suburu
- success
- sugar
- suicune
- summer
- sunflower
- sunshine
- super
- superhero
- superheroes
- superman
- supplies
- support
- supreme
- surrounded
- susu
- suwir
- swarovski
- sweater
- switch
- sword
- symbol
- symptoms
- system
- table
- tablet
- tablets
- tacoma
- tahu
- tahun
- takaran
- take
- talam
- talk
- tamil
- tank
- tanks
- tanpa
- tape
- target
- tassel
- tattoo
- taycan
- tdap
- teacher
- teachers
- teal
- team
- teasers
- technology
- teddy
- teflon
- tegal
- tell
- telo
- telur
- tempe
- tempeh
- template
- tenggiri
- tepung
- terang
- terasi
- terbaik
- terbaru
- terigu
- term
- test
- tetanus
- teton
- texas
- texture
- textured
- thailand
- thanksgiving
- theatre
- theme
- themed
- themes
- there
- thermos
- thinner
- thor
- thread
- three
- thrive
- throw
- thursday
- tiara
- tidak
- tier
- tiles
- timber
- timberlands
- time
- times
- tint
- tinta
- tintin
- tiny
- tips
- tiram
- tirupati
- titer
- today
- toddler
- toddlers
- tofu
- tokyo
- tomat
- tongkol
- touch
- tower
- townhouse
- toys
- tracing
- tracker
- tracking
- tradisional
- trailers
- trainer
- trainers
- transaction
- transfer
- transparent
- trap
- treatable
- tree
- trees
- trek
- trending
- tres
- trial
- trials
- trio
- triptych
- trolls
- tron
- trophy
- tropical
- truck
- true
- trump
- tuesday
- tulang
- tuna
- tunes
- turtles
- tutorial
- types
- typhoid
- typhus
- udaipur
- ugly
- ultra
- unban
- uncharted
- underwear
- ungkep
- unicorn
- unique
- unit
- united
- unlock
- unscramble
- untuk
- unvaccinated
- update
- updates
- uranium
- urap
- urdu
- user
- usmc
- vaccinating
- vaccination
- vaccinations
- vaccine
- vaccines
- vacuum
- value
- valve
- vans
- variable
- variables
- vector
- vegas
- vegetable
- vehicle
- velvet
- venice
- venkateswara
- venue
- verse
- version
- vert
- victorian
- video
- videos
- vietnam
- view
- views
- village
- vinci
- vintage
- vinyl
- viral
- virtual
- virus
- vishnu
- vision
- visit
- vocabulary
- vocational
- vogue
- volume
- vowel
- wali
- walik
- walkthrough
- wall
- wallpaper
- wallpapers
- walls
- walmart
- warm
- wars
- wash
- washable
- washington
- waste
- watch
- water
- watercolor
- watercolors
- waterfall
- waterpik
- wave
- waves
- wayfair
- wayne
- weaknesses
- wear
- wearing
- website
- websites
- wedding
- weed
- weirdest
- were
- west
- Western
- whale
- what
- when
- where
- which
- whipped
- whisky
- white
- whole
- wholesale
- whooping
- widescreen
- wieco
- wifi
- wikipedia
- wildflower
- wildlife
- will
- william
- williams
- window
- windows
- winter
- wireless
- wishes
- wisman
- with
- without
- wolf
- woman
- women
- womens
- wood
- woodblock
- wooden
- word
- wording
- words
- work
- working
- workouts
- works
- worksheet
- worksheets
- workshop
- world
- worldwide
- woven
- wrap
- wraps
- writing
- xmas
- yakiniku
- yang
- yantra
- year
- yeezy
- yeezys
- yellow
- york
- young
- your
- youth
- youtube
- yuno
- zearn
- zebra
- zigzag
- zika
- zombies
- zone
- zoro
